Catalytic reforming is a process which is born out the the necessity for high octane fuels which are used in transportation such as gasoline and aviation fuel.. This process takes the low octane straight run naphtha that is rich in naphthenes into high octane and low sulfur fuel which is used heavily in gasoline. Using this process a higher yield of gasoline can be achieved. In order to do this a few chemical reactions which increase the octane number of naphtha, convert the naphthenes to aromatics, and insomerize n-paraffins to i-paraffins must be completed. The catalysts used to bring these reactions to completion are generally platinum, palladium, or bimetallic formulation of platinum with Iridium or Rhenium with alumina. The process creates a large volume of hydrogen, which is a very valuable bi-product because it is in high demand for hydro-treating and hydro-cracking processes. The catalytic reformer is fed by the heavy naphtha line from the naphtha splitter. The naphtha splitter is fed by the straight run naphtha. The most desirable chemical reactions in the catalytic reforming process is dehydrogenation of naphthenes to aromatics, dehydroisomerization of alkyl-c5-naphthenes, Dehydrocyclization of n-paraffins to aromatics, and isomerization of n-alkanes to i-alkanes. These reactions increase the Research Octane Number (RON) from 75 to 110, 83 to 100, 0 to 110, and -19 to 90 respectively. All of these reactions except for the isomerization of n-alkanes produces hydrogen. If sufficient hydrogen is not produced coking my begin.
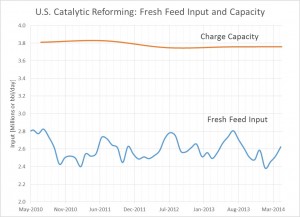
The catalytic reforming process is not without its limits. The United States and Europe limits the amount of benzene and other aromatics which can be found in gasoline. Because these compounds are carcinogenic and are harmful to the environment in many cases, catalytic reforming is limited. The high demand for high octane fuels and the hydrogen by product makes the process very valuable to refineries despite the production of aromatics.